search
Hands-on Experience That Deepens Your Faith
Glorify God by using science and technology to solve real-world research problems. You’ll define and answer significant questions about our world through a Christian lens. As a science major at Houghton, you’ll “Learn by doing,” thinking like a scientist and developing an appreciation for God’s creation and it’s infinite complexity.
Calling all deep thinkers, curious minds, researchers and problem solvers… we have a science major for you .
Take a look.
Biology Chemistry Physics
Biology Majors
Undergraduate Major & Minor – BA, BS
Undergraduate Major – BA, BS
Pre-Health Programs
Chemistry Majors
Undergraduate Major & Minor – BA, BS
Undergraduate Major – BA, BS
Physics Majors
Undergraduate Major & Minor – BA, BS
Natural Sciences at Houghton
Faculty Mentoring
Houghton’s experienced science faculty all have doctorates in their fields and are active scholars . They’ll partner with you and work side by side with you on research projects. You’ll find that Houghton’s Christian faculty care not only about your academic growth, but your emotional and spiritual growth as well .
Christ-Centered Education
One of the benefits of a Christian liberal arts education in the sciences is that you aren’t just exposed to learning about science. You are encouraged to deepen your faith as you increase your knowledge in your area of interest.
Experiential Learning
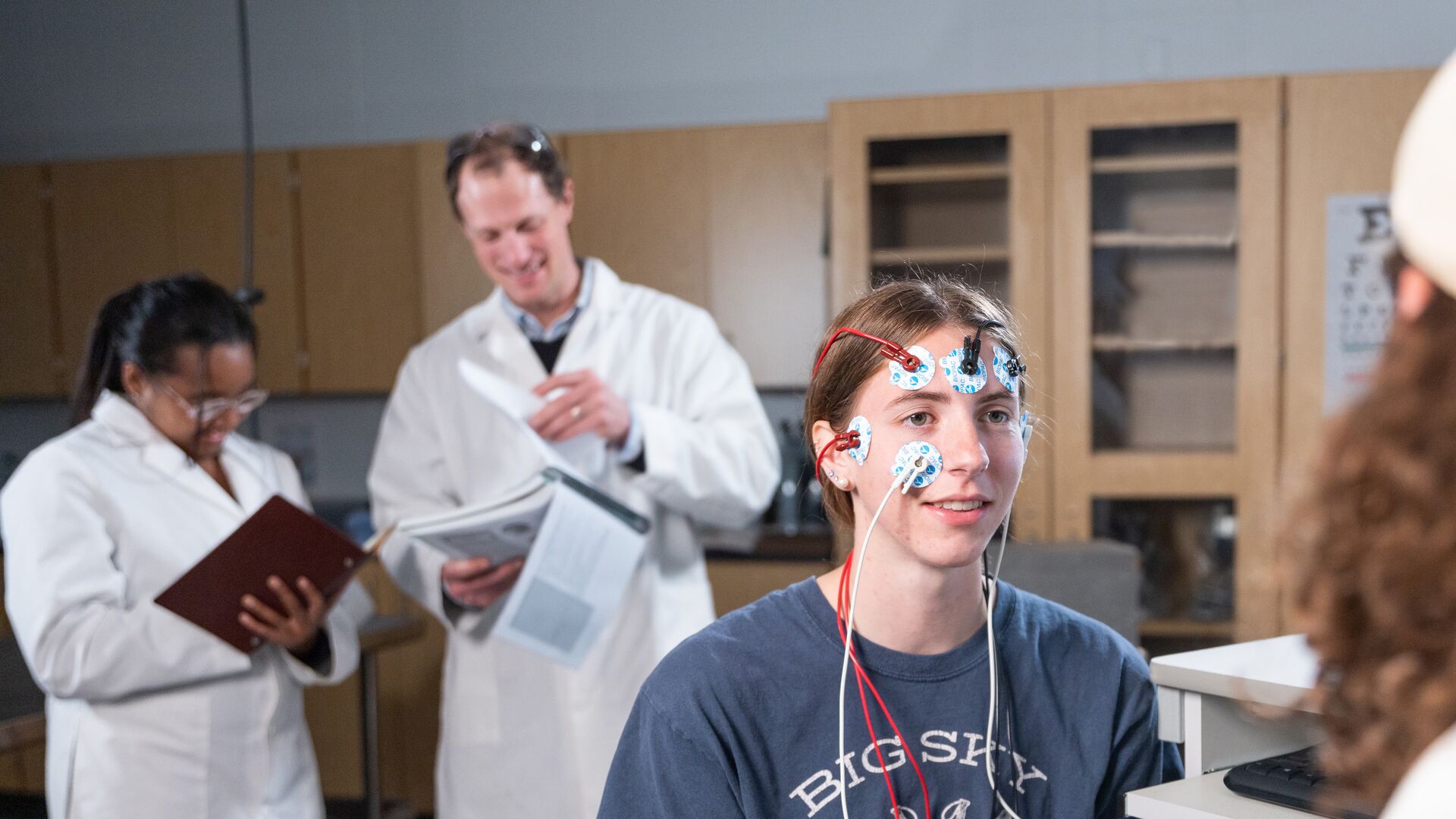
At Houghton, our faculty are committed to excellence in their teaching and research programs. You’ll find numerous opportunities to perform real-world research. You’ll have the opportunity to conduct, present, and publish peer-reviewed research in collaboration with faculty members. You can also work directly in the field through off-campus study, practicums, internships, and student/faculty research collaborations at the Shannon Summer Research Institute .
Explore the Shannon Summer Research Institute
State-of the-art Labs & Equipment
At Houghton, you’ll gain practical research and design experience in state-of the-art laboratories. The best way to learn is by doing and the science curriculum emphasizes practical laboratory experience. This means that you will be actively participating in studies and research using our professional research equipment and facilities, both at Houghton and other sites.
Explore Physics Labs Explore Biology Facilities & Equipment
Begin your college career with a hands-on research experience using a Drone for Remote Environmental Assessment and Monitoring (DREAM). Working with a team of peers, you will have the unique opportunity to design your own research project. Travel to Puerto Rico to deploy your DREAM on the tropical coastline or mountainous rainforest, gathering and assessing data to explore your research question.
Discover Science Honors
Meaningful Research
Adam Brown ’23 loves science and chose Houghton University because of the honors program and the opportunity to complete meaningful research as an undergrad. As a student, he has completed over 1200 hours of hands-on scientific research and conducted complicated experiments working side by side with classmates and professors. While he enjoyed his honors classes and the chance to develop his writing – his experiences with the Shannon Summer Research Institute and work with fellow physicists gave him the exciting opportunity to present his research at two national physics conferences. His love for learning, passion for STEM and the skills he has developed at Houghton have prepared him for graduate work and a future career in physics after graduation.
Explore Physics Student Research
David Brubaker
Assistant Professor of Biology, Director of Health Services, College Physician
585.567.9484
Send A Message
Health Center (Basement Gillette Hall), and Paine 318 Read Bio
Miriam Case
Academic Departments Coordinator for Paine Science Center
585.567.9280
Send A Message
Paine 219 Read Bio
Daniel Hammers
Assistant Professor of Biology
(585) 567-9274
Send A Message
Paine 306 Read Bio
Brandon Hoffman
Professor of Physics
585.567.9235
Send A Message
Paine 107 Read Bio
Jill Jordan
Dean of Natural Sciences & Mathematics; Associate Professor of Mathematics
585.567.9264
Send A Message
Paine 209C Read Bio
Eli Knapp
Professor of Ecology
585.567.9445
Send A Message
Chamberlain Center 406 Read Bio
Katrina Koehler
Assistant Professor of Physics
Send A Message
Paine 108 Read Bio
Paul Martino
Professor of Biochemistry
585.567.9300
Send A Message
Paine 420A Read Bio
Eden Palmer
Adjunct Professor of Exercise Science Read Bio
Jamie Potter
Professor of Biology; Director of Pre-Health Professions
585.567.9296
Send A Message
Paine 308 Read Bio
Ransom Poythress
Associate Professor of Biology
585.567.9279
Send A Message
Paine 307 Read Bio
John Rowley
Professor of Chemistry
585.567.9301
Send A Message
Paine 422 Read Bio
Babafemi Sorinolu
Visiting Assistant Professor of Computer Science
585.567.9259
Send A Message
Paine 209B Read Bio
Karen Torraca
Professor of Chemistry, Alan & Florence Johnson Distinguished Chair in Health Sciences
585.567.9383
Send A Message
Paine 420 Read Bio
Vanessa White
Adjunct Professor of Exercise Science, DPT Read Bio
Don Wilcox
Adjunct Assistant Professor of Biology
585.567.9293
Send A Message
Paine 315 Read Bio
Mark Yuly
Professor of Physics
585.567.9282
Send A Message
Paine 106 Read Bio
Load More
{"post_type":"staff","posts_per_page":18,"orderby":"last_word","order":"ASC","tax_query":[{"taxonomy":"staff-tax","field":"term_id","terms":["39"]}]}
[{"title":"Young Alumni Giving Society","link":"https:\/\/www.houghton.edu\/giving\/giving-societies\/young-alumni\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-231027-CampusFoliage-35.jpg","keywords":"giving, giving societies"},{"title":"Writing Major","link":"https:\/\/www.houghton.edu\/humanities\/writing-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/student-writing.jpg","keywords":"composition, prose, novelist, novel, writer, poet, story, book, writing, journalism, advertising, publishing, author, writing arts, literature, poetry, fiction, non-fiction, editor, communication specialist, technical writer, advocacy, freelance, blogging, blogger, magazine writer, reporting, content writer, editorial"},{"title":"Writing Center","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/writing-center\/","image":false,"keywords":"writing, writing center, writing support, writing help, writing process, outlining, drafting, revising, editing, papers, essay, academic support, academic success, student success"},{"title":"Worship Arts Major (BS)","link":"https:\/\/www.houghton.edu\/music\/worship-arts-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/05\/updated-images.jpg","keywords":"music, musical, musician, worship, composition, composer, writing music, aria, concerto, composing, worship, worship arts, music technology, songwriting, theology, biblical studies"},{"title":"Women's Choir","link":"https:\/\/www.houghton.edu\/music\/ensembles\/womens-choir\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/College-Choir-2019-Website-Header.jpg","keywords":"women's choir, women choir, choir, ensemble"},{"title":"Wind Ensemble","link":"https:\/\/www.houghton.edu\/music\/ensembles\/wind\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Wind-Ensemble-student-playing.jpg","keywords":"wind ensemble, ensemble"},{"title":"What We Believe","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/spiritual-life\/what-we-believe\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/Chapel-Worship.jpg","keywords":"beliefs, christian beliefs, biblical beliefs"},{"title":"Wellness Room","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/counseling\/wellness-room\/","image":false,"keywords":"Wellness, Wellness room, counseling"},{"title":"Volleyball Summer Camp","link":"https:\/\/www.houghton.edu\/summer-camps\/volleyball-camp\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/02\/Volleyball-2022-Action-Website-Header.jpg","keywords":"volleyball, camp, summer"},{"title":"Visitor Resources","link":"https:\/\/www.houghton.edu\/visit\/visitor-resources\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/About-Aerial_1.jpg","keywords":"accommodations, local hotels, local dining, where to eat by campus, restaurants"},{"title":"Visit Campus","link":"https:\/\/www.houghton.edu\/visit\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/10\/Houghton-Universtiy-230526-Campus-in-Summer-41.jpg","keywords":"visit, visiting, campus, tour, virtual event, v, vi,"},{"title":"Veterans Benefits","link":"https:\/\/www.houghton.edu\/military-resources\/veterans-benefits\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/04\/Army-ROTC-in-Fatigues.jpg","keywords":"military benefits, military, army, rotc, veterans"},{"title":"University Post Office","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/post-office\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Campus-Center.jpg","keywords":"packages, mail, student ID"},{"title":"Undergraduate Music Auditions","link":"https:\/\/www.houghton.edu\/music\/undergraduate-auditions\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Symphony-Orchestra-violin.jpg","keywords":"music auditions, audition"},{"title":"Undergraduate","link":"https:\/\/www.houghton.edu\/undergraduate\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/Houghton_students_campus_swing.jpg","keywords":"undergraduate, undergrad, bachelor, houghton, ny, campus, residential, liberal arts"},{"title":"Types of Aid","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/types-of-aid\/","image":false,"keywords":"financial aid"},{"title":"Tuition & Aid\u2014Online","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/tuition-aid\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/student_online.jpg","keywords":"tuition, online, price, cost, scholarship, financial aid, aid, award,"},{"title":"Tuition & Aid\u2014MBA","link":"https:\/\/www.houghton.edu\/graduate\/master-of-business-administration\/tuition-aid\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/08\/MBA-Conference-Room.jpg","keywords":"MBA, financial aid, tuition, loans"},{"title":"Tuition & Aid\u2014Houghton, NY","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/05\/Commencement-Processional.jpg","keywords":"tuition, reset, price, cost, scholarship, financial aid, aid, award,"},{"title":"Trips, Outings and Experiences","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/epic-adventures\/trips\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/kayak-scaled.jpg","keywords":"EPIC adventures, outdoor trips, sunset paddle, kayaking, canoeing, hiking, camping, zipline"},{"title":"Transfer Students","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/transfer-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/02\/Houghton-University-230825-MoveInDay77-1.webp","keywords":"transfer students, undergraduate, houghton, ny"},{"title":"Transcripts","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/transcripts\/","image":false,"keywords":"registrar"},{"title":"Townhouses","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/residence-life\/townhouses\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/02\/Houghton_230227-Townhouse-Exterior-6.jpg","keywords":"townhouse, townhouses, residence life, residence halls, living on campus"},{"title":"Title IX and Related Compliance: Safety and Equity at Houghton University","link":"https:\/\/www.houghton.edu\/current-students\/title-ix-and-related-compliance-safety-and-equity-at-houghton-college\/","image":false,"keywords":"title IX, compliance, equity, policy, safety"},{"title":"Three-Year Option","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/three-year-option\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Computer-Science-2016-Website-Header-1.jpg","keywords":"three-year option, 3 year option, early graduation, accelerated graduation"},{"title":"The Houghton Promise","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/houghton-promise\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/02\/Houghton_221020-Streaming_Student_Sitting.jpg","keywords":"promise, houghton promise, guarantee, houghton guarantee, bonus year"},{"title":"The Highlanders Printing Press","link":"https:\/\/www.houghton.edu\/the-highlanders-printing-press\/","image":false,"keywords":"Quick print"},{"title":"The Highlander Shop","link":"https:\/\/www.houghton.edu\/the-highlander-shop\/","image":false,"keywords":"book store, campus store, textbooks, store, highlander shop, shop,"},{"title":"Textbooks","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/course-selection\/textbooks\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/2022-Textbooks.jpg","keywords":"books, bookstore, online bookstore"},{"title":"Testing Policy","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/apply\/testing-policy\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/student_highlighting.jpg","keywords":"testing policy, SAT, ACT, test free, test optional, standardized tests, admission, apply, applying"},{"title":"Tennis Summer Camp","link":"https:\/\/www.houghton.edu\/summer-camps\/tennis-camp\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/11\/220923-Tennis-MarceloGarcia-11.jpg","keywords":"tennis, camp, summer"},{"title":"Technology, Email & Student ID","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/technology-email-student-id\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/laptop.jpg","keywords":"technology, computers, laptops, student ID, student email"},{"title":"Technology Services","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/technology-services\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/Investment-Center.jpg","keywords":"helpdesk, telecom, computer support"},{"title":"Symphony Orchestra","link":"https:\/\/www.houghton.edu\/music\/ensembles\/symphony-orchestra\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Prism.jpg","keywords":"symphony orchestra, strings"},{"title":"Summer Research Internships","link":"https:\/\/www.houghton.edu\/natural-sciences\/summer-programs\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/10\/phsyics_student_1.jpg","keywords":"faculty research, student research, summer research, nanotechnology, nuclear, physics, artificial intelligence, AI, fusion, metrology, shannon summer institute"},{"title":"Summer Online Courses","link":"https:\/\/www.houghton.edu\/online\/online-courses\/summer-online-courses\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/nature_white-flowers.jpg","keywords":"summer online, online courses, online course offerings, summer online courses, summer online course offerings, summer courses"},{"title":"Summer at Houghton","link":"https:\/\/www.houghton.edu\/summer-camps\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/10\/Houghton-University-22-Girls-Soccer-Camp.jpg","keywords":"Camps, Athletic camps, Basketball camp, Soccer camp, Volleyball camp, Tennis camp, Equestrian camp, Summer camps, Children\u2019s camps, summer camp"},{"title":"Studio Art Major","link":"https:\/\/www.houghton.edu\/art\/studio-art-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/11\/Art-Pottery.jpg","keywords":"art, bfa, fine arts, studio, studio art, artist, fine artist, sculpting, sculptor, printer, printmaking, book making, ceramics, pottery, drawing, fiber, graphic design, designer, painting, painter, photography, photographer, art, drawing, painting, sculpture, 3d design, pottery, ceramics, lithography, photography, film, BFA, fine arts, studio, art history, 3d art, woodwork, carpentry, book arts, fiber arts, print making, illustrator, art teacher, art education, potter,"},{"title":"Student Services","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/08\/Houghton-University-230526-Campus-in-Summer-15.jpg","keywords":"college post office, safety and security, safety, student health services, health services, medical center, payroll, student payroll, technology services, helpdesk, student services, student life"},{"title":"Student Payroll","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/payroll\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/passportday-houghton.jpg","keywords":"student payroll, on-campus employment, campus jobs, student jobs, working on-campus, working, jobs"},{"title":"Student Pay Schedule","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/payroll\/student-pay-schedule\/","image":false,"keywords":"student payroll"},{"title":"Student Life","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/11\/2022-Textbook-Giveaway_1.jpg","keywords":"campus life, student life, clubs, organization, sports, recreation, culture, arts"},{"title":"Student Life","link":"https:\/\/www.houghton.edu\/current-students\/student-engagement\/","image":false,"keywords":"student life, campus life, clubs, clubs and organizations"},{"title":"Student Health Services","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/health-services\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Gillette-Hall.jpg","keywords":"student health services, health, doctor, nurse, physician, doctor's office, physicals, health center"},{"title":"Student Employment in Admission Office","link":"https:\/\/www.houghton.edu\/admission-student-employment\/","image":false,"keywords":"student employment, on-campus job, campus jobs, campus employment"},{"title":"Student Accounts Policies","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/resources-faqs\/student-accounts-policies\/","image":false,"keywords":"late fees, financial holds, appeal, appeals, refund policy"},{"title":"Sport, Recreation and Wellness Management Major","link":"https:\/\/www.houghton.edu\/health\/sport-recreation-wellness-management-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Martial-Arts.jpg","keywords":"sports management, recreation management, sport, recreation, wellness, wellness management, fitness, recreation and wellness, coaching, athletic training, sports ministry, camp director, physical education, athletic administration, athletic director, AD, sports management,"},{"title":"Sponsor Houghton's Summer Christian School Symposium","link":"https:\/\/www.houghton.edu\/partners\/sponsor\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-220824-Move-In-06.jpg","keywords":"partners, summer symposium. Christian school partners"},{"title":"Spiritual Life","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/spiritual-life\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-230115-KOIN-5-Header.jpg","keywords":"chapel, spiritual life, faith, christian, bible, christianity, spiritual formation, formation, church, evangelical, evangelicalism"},{"title":"Social Sciences","link":"https:\/\/www.houghton.edu\/social-sciences\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/psych-lab.jpg","keywords":"psychology, behavioral neuroscience, criminal justice, pre-art therapy, art therapy, pre-law, law"},{"title":"Soccer Summer Camp","link":"https:\/\/www.houghton.edu\/summer-camps\/soccer-camp\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Womens-Soccer-2022-Action-Website-Header-1.jpg","keywords":"soccer camp, summer, summer camps, western NY, kids camps"},{"title":"Share Your Updates","link":"https:\/\/www.houghton.edu\/alumni\/share-your-updates\/","image":false,"keywords":"alumni, alumnus, alumna, alum, update, news"},{"title":"Share Your Feedback","link":"https:\/\/www.houghton.edu\/share-your-feedback\/","image":false,"keywords":"search, feedback"},{"title":"Shannon Summer Research Institute","link":"https:\/\/www.houghton.edu\/about\/centers-and-institutes\/summer-research-institute\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/10\/Physics_classroom_2.jpg","keywords":"summer research, shannon summer research institute,"},{"title":"Security and Fire Safety Information","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/safety-security\/security-fire-safety\/","image":false,"keywords":"fire safety, security"},{"title":"Sculpture","link":"https:\/\/www.houghton.edu\/art\/studio-art-major\/sculpture\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Ceramics-Class-Website-Header.jpg","keywords":"art, fine arts, studio, studio art, artist, fine artist, printer, BFA, art history, 3d art, art teacher, art education, sculpture, woodworking, metalwork, jewelry, art installation, 3D digital design, 3D printing, figure sculpture, stone carving, clay,"},{"title":"Science Honors","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/honors\/science\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/06\/Science-Honors-aerial-panorama-of-el-morro-fort-and-san-juan-pue-2021-08-26-16-22-39-utc-1.jpg","keywords":"honors, science, Puerto Rico"},{"title":"Scholarships and Grants","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/types-of-aid\/scholarships-grants\/","image":false,"keywords":"scholarships, grants, financial aid"},{"title":"Rothenbuhler Hall","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/residence-life\/rothenbuhler-hall\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/02\/230208-Residence-Hall-Exterior-Roth-1.jpg","keywords":"residence hall, housing, residence life, living on campus, rooms"},{"title":"Rights and Responsibilities of Financial Aid Recipients","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/resources-faqs\/policies\/rights-responsibilities\/","image":false,"keywords":"rights and responsibilities, financial aid, aid"},{"title":"Rhetorical Communication Concentration","link":"https:\/\/www.houghton.edu\/business\/communication-major\/rhetorical-communication\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Business-Class-2019-presenting.jpg","keywords":"rhetorical communication, communications, public relations, journalism, broadcasting, government service, law, business, teaching, ministry"},{"title":"Returning Students","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/returning-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/220824-Move-In-Luckey-12.jpg","keywords":"returning students, admission, undergraduate"},{"title":"Resources and Scholarship Information","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/off-campus-studies\/resources\/","image":false,"keywords":"scholarships off-campus study, scholarships off-campus studies"},{"title":"Residence Life & Housing","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/residence-life\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/02\/Houghton_230214-Girl-Dorm-Room-8.jpg","keywords":"housing, meal plans, mayterm, room draw, dorm, living on campus, residence halls, dining, resident hall"},{"title":"Request Information","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/request-info\/","image":false,"keywords":"rfi, request for information, request information, info form"},{"title":"Request Info","link":"https:\/\/www.houghton.edu\/request-info\/","image":false,"keywords":"info, information, request, get info, learn more,"},{"title":"Replacement Diploma","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/replacement-diploma\/","image":false,"keywords":"diploma, replacement"},{"title":"Registered Early College Students","link":"https:\/\/www.houghton.edu\/early-college\/registered-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/02\/Students-Computers-Online.jpg","keywords":"early college, dual enrollment"},{"title":"Psychology Major","link":"https:\/\/www.houghton.edu\/social-sciences\/psychology-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/01\/Psychology-Lab_Cap-Website-Header.jpg","keywords":"psychology, counseling, psychologist, counselor, psychiatrist, psychiatry, mental health, neuroscience, clinical, diversity,"},{"title":"Psychology Internships","link":"https:\/\/www.houghton.edu\/social-sciences\/psychology-major\/internships\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/psychology_classroom.jpg","keywords":"psychology interships"},{"title":"Psychology","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/programs\/psychology\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/ONL-Psychology-Header.jpg","keywords":"psychology, counseling, psychologist, counselor, psychiatrist, psychiatry, mental health, online psychology"},{"title":"President's Advisory Board","link":"https:\/\/www.houghton.edu\/about\/president\/presidents-advisory-board\/","image":false,"keywords":"president, advisory board"},{"title":"President","link":"https:\/\/www.houghton.edu\/about\/president\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/07\/Lewis-Wayne-Quad.jpg","keywords":"president, lewis, wayne"},{"title":"Pre-Veterinary","link":"https:\/\/www.houghton.edu\/health\/pre-veterinary\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/dog.jpg","keywords":"veterinarian, veterinary, pre-veterinary, dog, cat, horse, equestrian, bird, animal, zoology, medicine, pre-health, healthcare, health, paraveterinary,"},{"title":"Pre-Physical Therapy","link":"https:\/\/www.houghton.edu\/health\/pre-physical-therapy\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/biology_lab.jpg","keywords":"physical therapy, pt, pre-pt, pre-physical therapy, therapy, medicine, pre-health, healthcare, health, rehab, rehabilitate,"},{"title":"Pre-Pharmacy","link":"https:\/\/www.houghton.edu\/health\/pre-pharmacy\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/student_biology.jpg","keywords":"pharmacy, pre-pharmacy, pharmaceutical, pharmacist, prescription, medicine, pre-health, healthcare, health, rx,"},{"title":"Pre-Nursing","link":"https:\/\/www.houghton.edu\/health\/pre-nursing\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/student-wrapping-bandage.jpg","keywords":"nurse, nursing, pre-nursing, medicine, healthcare, health, RN, medical, BSN, registered nurse"},{"title":"Pre-Music Therapy","link":"https:\/\/www.houghton.edu\/music\/pre-music-therapy\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/09\/Houghton-University-guitar-1.jpg","keywords":"music therapy, special education, health care, music therapist"},{"title":"Pre-Med Requirements","link":"https:\/\/www.houghton.edu\/health\/pre-med\/pre-med-requirements\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/biology-students.jpg","keywords":"pre-med requirements, pre-med, medical school"},{"title":"Pre-Med","link":"https:\/\/www.houghton.edu\/health\/pre-med\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/biology-students.jpg","keywords":"medicine, pre-medicine, doctor, m.d., md, medical doctor, pre-health, healthcare, health, osteopathic, pre-med,"},{"title":"Pre-Law","link":"https:\/\/www.houghton.edu\/humanities\/pre-law\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/04\/Business-Class-2019-Website-Header-1-e1650397481119.jpg","keywords":"pre-law, law, lawyer, judge, judicial, justice,"},{"title":"Pre-Art Therapy","link":"https:\/\/www.houghton.edu\/social-sciences\/pre-art-therapy\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/student-artwork-1280.jpg","keywords":"art therapy, art, therapy, counseling, psychology, mental health"},{"title":"Politics & Philosophy Major","link":"https:\/\/www.houghton.edu\/humanities\/politics-philosophy-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Business-Class-2019-Website-Header.jpg","keywords":"politics, political science, philosophy, economics, policy, public policy, law, pre-law, PPE"},{"title":"Physics, Computer Science and Engineering Laboratories & Equipment","link":"https:\/\/www.houghton.edu\/natural-sciences\/physics-facilities\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/10\/HoughtonUniversity_PhysicsLab_1.jpg","keywords":"engineering, physics, lab, facilities, equipment"},{"title":"Physics Research","link":"https:\/\/www.houghton.edu\/natural-sciences\/physics-research\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Physics_bridge.jpg","keywords":"physics research, project lab, shannon summer research institute, science honors, undergraduate research, physics"},{"title":"Physics Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/physics-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/10\/HoughtonUniversity_PhysicsLab.jpg","keywords":"physics, research scientist, engineering, energy, technology, physical science, teacher, researcher, R&D, development, research and development, engineer, programmer, developer, accelerator operator, engineering design"},{"title":"Photography","link":"https:\/\/www.houghton.edu\/art\/studio-art-major\/photography\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Photography-2019-Website-Header.jpg","keywords":"art, fine arts, studio, studio art, artist, fine artist, BFA, art history, photography, lighting, moving image, darkroom, visual communication, visual culture, photo, digital photography, travel photography, commercial photography, photographer art teacher, art education"},{"title":"Personal Essay","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/apply\/personal-essay\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/Fearless-JenniferPage.jpg","keywords":"personal essay, statement, applying, application"},{"title":"Performance, M.M.","link":"https:\/\/www.houghton.edu\/graduate\/music\/programs\/performance-m-m\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Orchestra-trombone.jpg","keywords":"music, musical, worship, lesson, audition, choir, chorus, band, wind ensemble, orchestra, symphony, strings, brass, woodwind, percussion, drums, voice, piano, organ, guitar, flute, clarinet, saxophone, oboe, bassoon, trumpet, horn, trombone, euphonium, baritone, tuba, drums, violin, viola, cello, bass, soprano, alto, tenor, performance, collaborative piano, jazz, A cappella, performances, concert, aria, concerto competition, recital, harpsichord, music theory, music history, global music, world music, master's in music, music master's, graduate music,"},{"title":"Paying Your Bill","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/paying-your-bill\/","image":false,"keywords":"bill, pay, new students, financial services"},{"title":"Partners","link":"https:\/\/www.houghton.edu\/partners\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Campus-Bridge.jpg","keywords":"early college, dual enrollment, Christian partner network, high schools, private schools, college prep"},{"title":"Parking Ticket Appeal","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/safety-security\/parking-permit\/ticket-appeal\/","image":false,"keywords":"parking, permit, registration, appeal, safety, security"},{"title":"Parking Permit Registration","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/safety-security\/parking-permit\/registration\/","image":false,"keywords":"parking, registration, permit"},{"title":"Parking Permit Information","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/safety-security\/parking-permit\/","image":false,"keywords":"parking, permit, registration, appeal"},{"title":"Parents & Families - International Student","link":"https:\/\/www.houghton.edu\/parents-families\/international-student\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/03\/Houghton_University_230315-MulticulturalMediaDay.jpg","keywords":"international students, parents, families"},{"title":"Parents & Families - Current Student","link":"https:\/\/www.houghton.edu\/parents-families\/current-student\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-220824-Move-In-07-1.jpg","keywords":"parents, family, resource, guardians"},{"title":"Parents & Families - Accepted Student","link":"https:\/\/www.houghton.edu\/parents-families\/accepted-student\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-230920-Bookshop_Buckethat-07.jpg","keywords":"accepted student, parents, families"},{"title":"Parents & Families","link":"https:\/\/www.houghton.edu\/parents-families\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-230513-Commencement-09.jpg","keywords":"parents, family, resource, guardians"},{"title":"Painting","link":"https:\/\/www.houghton.edu\/art\/studio-art-major\/painting\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Painting-2019-Website-Header.jpg","keywords":"BFA, studio art, art, painting, oil painting, acrylic painting, mixed media, watercolor, pastel, drawing, art education"},{"title":"Packing List and Residence Hall Supplies","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/new-student-orientation-and-information\/packing-list-and-residence-hall-supplies\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/students-residencehalls.jpg","keywords":"packing list, residence life, dorm, living on campus, gillette, lambein, rothenbuhler, roommate"},{"title":"Other Opportunities","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/off-campus-studies\/opportunities\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/London-Honors-2018.jpg","keywords":"off campus, abroad"},{"title":"Ortlip Gallery","link":"https:\/\/www.houghton.edu\/art\/ortlip-gallery\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/Ortlip-Gallery-2019-Website-Header.jpg","keywords":"ortlip gallery, art, gallery, contemporary art, artists, art show"},{"title":"Organizational Management Major","link":"https:\/\/www.houghton.edu\/business\/organizational-management-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Business-Class-Houghton-University.jpg","keywords":"business, management, leadership, organization, project management"},{"title":"Organizational Management","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/programs\/organizational-management\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/Business-Investment-Center-1.jpg","keywords":"business, management, leadership, organization, online business, systems, HRIS, employee relations,"},{"title":"OPT Employment","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/intercultural-programs\/employment-non-citizen\/opt\/","image":false,"keywords":"optional practical training, international students"},{"title":"Online\u2014Undergraduate","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/student_online.jpg","keywords":"degree completion, bachelors completion, adult, professional, evening class,"},{"title":"Online Courses","link":"https:\/\/www.houghton.edu\/online\/online-courses\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/student_online.jpg","keywords":"online, online courses, online course offerings"},{"title":"Online","link":"https:\/\/www.houghton.edu\/online\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/05\/Sagi-Melanie-5-Website-Header.jpg","keywords":"online, graduate, adult, professional, online degrees, online programs,"},{"title":"Office of Vocation and Calling","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/vocation-and-calling\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/02\/Houghton-University-220518-VOCA-InterviewRoom-6.webp","keywords":"vocation and calling, career services, career development, job search, internships"},{"title":"Off-Campus Studies","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/off-campus-studies\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Houghton-Tanzania.jpg","keywords":"off campus, study abroad, abroad, international, travel, semester abroad, mayterm,"},{"title":"NYS High School Equivalency Diploma","link":"https:\/\/www.houghton.edu\/early-college\/diploma\/","image":false,"keywords":"early college, dual enrollment, diploma, high school equivalency, college credit"},{"title":"New York State Certification Information","link":"https:\/\/www.houghton.edu\/education\/certification\/","image":false,"keywords":"teaching certification, initial certification, nys certification"},{"title":"New York State Aid","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/types-of-aid\/nys-aid\/","image":false,"keywords":"financial aid, new york state aid, nys, ny, scholarships, grants"},{"title":"New Students","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghhton-University-230825-MoveInDay01.jpg","keywords":"new students, checklist, verification, accepted students"},{"title":"New Student Orientation and Information","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/new-student-orientation-and-information\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/220824-Move-In-Luckey-07.jpg","keywords":"move in, arrival, orientation"},{"title":"Neuroscience Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/neuroscience-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-230428-Biopac-2.jpg","keywords":"neuroscience, science, biology, psychology, natural sciences, social sciences, labs"},{"title":"Natural Sciences","link":"https:\/\/www.houghton.edu\/natural-sciences\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-University-Fearless-Oscar-Palacios-Bio-2.jpg","keywords":"biology, biomedical sciences, environmental science, exercise science, neuroscience, chemistry, biochemistry, physics, applied physics, research"},{"title":"National Recognition","link":"https:\/\/www.houghton.edu\/about\/national-recognition\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/09\/Houghton-Recognitions-Header.jpg","keywords":"recognitions, ranking, us news, best college"},{"title":"National Association for Music Education","link":"https:\/\/www.houghton.edu\/music\/nafme\/","image":false,"keywords":"nafme, national association, music"},{"title":"Music, M.A.","link":"https:\/\/www.houghton.edu\/graduate\/music\/programs\/music-m-a\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/violinist_mirror.jpg","keywords":"music, musical, worship, lesson, audition, music theory, music history, global music, world music, master's in music, music master's, graduate music,"},{"title":"Music with Non-Music Elective Studies Major (BMus)","link":"https:\/\/www.houghton.edu\/music\/music-non-music-elective-studies-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Music.jpg","keywords":"music, musical, worship, lesson, audition, music theory, music history, global music, world music,"},{"title":"Music Performance Major (BMus)","link":"https:\/\/www.houghton.edu\/music\/music-performance-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/11\/Concert-Violinist.jpg","keywords":"music, music performance, musical, worship, lesson, audition, choir, chorus, band, wind ensemble, orchestra, symphony, strings, brass, woodwind, percussion, drums, voice, piano, organ, guitar, flute, clarinet, saxophone, oboe, bassoon, trumpet, horn, trombone, euphonium, baritone, tuba, drums, violin, viola, cello, bass, soprano, alto, tenor, performance, collaborative piano, jazz, A cappella, performances, concert, aria, concerto competition, recital, harpsichord,"},{"title":"Music Major (BA)","link":"https:\/\/www.houghton.edu\/music\/music-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Music-Theory-Class.jpg","keywords":"music, musical, worship, lesson, audition, music theory, music history, global music, world music,"},{"title":"Music Industry Major (BS)","link":"https:\/\/www.houghton.edu\/music\/music-industry-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/music-industry-student.jpg","keywords":"music, musical, worship, lesson, audition, music industry, film music, recording studio, studio, recording artist, album, recording, production, management, broadcast, sound, tour manager, Nashville"},{"title":"Music Facilities & Equipment","link":"https:\/\/www.houghton.edu\/music\/facilities\/","image":false,"keywords":"music facilities, instruments"},{"title":"Music Education Major (BMus)","link":"https:\/\/www.houghton.edu\/music\/music-education-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Music-conducting.jpg","keywords":"music, musical, worship, lesson, audition, music education, music pedagogy, teaching music, music teacher, teacher, choir teacher, band teacher, choir, band, orchestra"},{"title":"Music Composition Major (BMus)","link":"https:\/\/www.houghton.edu\/music\/music-composition-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Music-Video-Shoot.jpg","keywords":"music, musical, worship, composition, composer, writing music, aria, concerto, film music, lesson, audition, film-scoring, orchestrating, publishing, music administration, composing"},{"title":"Music","link":"https:\/\/www.houghton.edu\/graduate\/music\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/05\/College-Choir-2019-Website-Header.jpg","keywords":"graduate, music, grad"},{"title":"Music","link":"https:\/\/www.houghton.edu\/music\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-230420-FEARLESS-Rachel-Huchthausen-7.jpg","keywords":"music, music major, music industry, composition, performance, worship arts, graduate programs, ensembles, lyric theatre, music staff"},{"title":"Ministry","link":"https:\/\/www.houghton.edu\/ministry\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-University-230312-KOIN-4.jpg","keywords":"ministry, bible, theology, worship arts, ministry leader, pastor, preacher, missionary, youth pastor, worship leader"},{"title":"Military Resources","link":"https:\/\/www.houghton.edu\/military-resources\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/11\/Fearless-KelseyVillatoro-4.jpg","keywords":"military resources, military friendly"},{"title":"Men's Choir","link":"https:\/\/www.houghton.edu\/music\/ensembles\/mens-choir\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/College-Choir-mens.jpg","keywords":"men's choir, choir, singing"},{"title":"Media Arts & Visual Communication Concentration","link":"https:\/\/www.houghton.edu\/business\/communication-major\/media-arts-visual-communication\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/04\/Houghton-University-240419-MacLab-02.webp","keywords":"media arts, communication, communications, visual communication, digital arts, animation, graphic design, photography"},{"title":"Mathematics Major","link":"https:\/\/www.houghton.edu\/math-science\/mathematics-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/04\/Math-2016-Website-Header-2.jpg","keywords":"math, mathematics, mathematician, statistics, statistician, actuary, actuarial, calculus, analyst, problem solving, data science,"},{"title":"Mathematical Sciences","link":"https:\/\/www.houghton.edu\/math-science\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-University-Fearless-Haley.jpg","keywords":"math, mathematics, statistics, computer science, data science, physics, applied physics"},{"title":"Math Student Research","link":"https:\/\/www.houghton.edu\/math-science\/math-research\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Math-Class.jpg","keywords":"mathematics, math, research"},{"title":"Master of Business Administration","link":"https:\/\/www.houghton.edu\/graduate\/master-of-business-administration\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/08\/MBA-Conference-Room.jpg","keywords":"master of business administration, mba, business, finance, leadership, management, ceo, cfo, cmo, executive, online MBA, christian MBA program"},{"title":"Majors & Programs","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/04\/Math-2016-Website-Header-3.jpg","keywords":"majors, programs, degrees, undergraduate, academics"},{"title":"Loans","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/types-of-aid\/loans\/","image":false,"keywords":"loans, financial aid"},{"title":"Loans","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/paying-your-bill\/loans\/","image":false,"keywords":"federal direct loans, federal perkins loan, federal PLUS loan, private loan, private loans, preferred lenders"},{"title":"Literacy Education, MSEd","link":"https:\/\/www.houghton.edu\/graduate\/literacy-education\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/01\/MSED-Teacher.jpg","keywords":"literacy, education, teacher, professional teacher certification, professional certification, certification, reading, educator, master's of education, master of education, m.ed.,"},{"title":"Library","link":"https:\/\/www.houghton.edu\/library\/","image":false,"keywords":"library"},{"title":"Lambein Hall","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/residence-life\/lambein-hall\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/09\/Houghton-University-230526-Campus-in-Summer-7-Lambein.jpg","keywords":"residence hall, housing, residence life, living on campus, rooms"},{"title":"Kern Scholarship Program for Ministry Leaders","link":"https:\/\/www.houghton.edu\/ministry\/bible-theology-major\/kern-scholarship-program\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/09\/Houghton-University-Spiritual-Life-hymns.jpg","keywords":"scholarship, ministry, ministry leader"},{"title":"Jazz Ensemble","link":"https:\/\/www.houghton.edu\/music\/ensembles\/jazz\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/music-Jazz.jpg","keywords":"jazz, ensemble, ensembles, big band"},{"title":"James S. Luckey Society","link":"https:\/\/www.houghton.edu\/giving\/giving-societies\/james-s-luckey-society\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/10\/J-Luckey-on-steps_corrected.jpg","keywords":"james luckey, luckey, giving, giving society"},{"title":"Internships","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/vocation-and-calling\/internships\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/Fearless-Ella-Osuzoka.jpg","keywords":"internships, internship, office of vocation and calling, voca"},{"title":"International Students, MKs and TCKS","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/international-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/03\/Houghton_University_230315-MulticulturalMediaDay.jpg","keywords":"international students, admission, applying, enrollment, undergrad, houghton, ny"},{"title":"International Payments","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/paying-your-bill\/international\/","image":false,"keywords":"international, tuition, paying tuition, flywire"},{"title":"International Business Institute","link":"https:\/\/www.houghton.edu\/business\/international-business-institute\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Investment-Center-Houghton.jpg","keywords":"international business, overseas program, off-campus, abroad"},{"title":"Intercultural Student Programs","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/intercultural-programs\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/03\/Houghton_University_230315-MulticulturalMediaDay.jpg","keywords":"international students, mck, tk, intercultural programming, interconnect, social security card, visa, us taxes, NY driver's license"},{"title":"InterCare","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/intercultural-programs\/intercare\/","image":false,"keywords":"international students, mck, tk, intercultural students, intercultural student program"},{"title":"Instrumental Ensemble Auditions","link":"https:\/\/www.houghton.edu\/music\/ensembles\/instrumental-auditions\/","image":false,"keywords":"auditions, ensemble, music"},{"title":"Inclusive Childhood Education Major","link":"https:\/\/www.houghton.edu\/education\/inclusive-childhood-education-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/02\/Houghton-University-Literacy-Day-2.webp","keywords":"inclusive childhood, education, teacher, elementary, childhood education, special education, SPED, educator"},{"title":"Inclusive Adolescence Education Major","link":"https:\/\/www.houghton.edu\/education\/inclusive-adolescence-education-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Student-Teaching-adjusted.jpg","keywords":"inclusive adolescence, education, teacher, teaching, certification, high school, SPED, educator, education"},{"title":"Impact and Outcome Measures","link":"https:\/\/www.houghton.edu\/education\/impact-outcome-measures\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/student-portrait-math-classroom.jpg","keywords":"teaching program, education program, impact and outcome measures, teacher education program"},{"title":"Humanities","link":"https:\/\/www.houghton.edu\/humanities\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-University-230315-MulticulturalMediaDay-Zambia-1.jpg","keywords":"Global Studies, History, English, Writing, Politics, Philosophy, Pre-Law, literature"},{"title":"Human Resources Management Major","link":"https:\/\/www.houghton.edu\/business\/human-resources-management-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/Business-Students-Class.jpg","keywords":"human resources, hr, business, labor law, personnel management, diversity consultant, consulting, employee relations, manager, industrial relations, labor policy analyst, non-profit, recruiter, staff coordinator, union organizer, payroll coordinator, compensation & benefits manager, compliance officer"},{"title":"Human Resources Management","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/programs\/human-resources-management\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-Sagi-Melanie-4.jpg","keywords":"business, management, hr, online degree"},{"title":"Houghton's Opportunities","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/off-campus-studies\/houghtons-opportunities\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/Big-Ben-18-Website-Header.jpg","keywords":"off-campus studies, abroad, london honors, highlander wilderness, mayterm"},{"title":"Houghton's 4+1 Program","link":"https:\/\/www.houghton.edu\/graduate\/master-of-business-administration\/houghtons-41-program\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/12\/HoughtonUniversity_Business-Class.jpg","keywords":"MBA, online MBA, 4+1, five year, early graduation, master's, business"},{"title":"Houghton University","link":"https:\/\/www.houghton.edu\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/houghton_university_commencement.jpg","keywords":"houghton university"},{"title":"Houghton Trail Map","link":"https:\/\/www.houghton.edu\/visit\/trail-map\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Houghton_Forest.jpg","keywords":"trails, trail, trail map, forest, woods"},{"title":"Houghton Magazine\u2014Winter 2024","link":"https:\/\/www.houghton.edu\/alumni\/houghton-magazine\/winter-2024\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/231206-StrangelyWarmed.webp","keywords":"magazine, alumni magazine"},{"title":"Houghton Magazine\u2014Summer 2023","link":"https:\/\/www.houghton.edu\/alumni\/houghton-magazine\/summer-23\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-Universtiy-Cambodia-Gibbon.jpg","keywords":"magazine, alumni magazine"},{"title":"Houghton Magazine\u2014Fall 2021","link":"https:\/\/www.houghton.edu\/alumni\/houghton-magazine\/houghton-magazine-fall-2021\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/President_Lewis_purpletie.jpg","keywords":"magazine, alumni magazine"},{"title":"Houghton Magazine","link":"https:\/\/www.houghton.edu\/alumni\/houghton-magazine\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/231206-StrangelyWarmed.webp","keywords":"magazine, milieu, alumni magazine,"},{"title":"Houghton Consulting Group","link":"https:\/\/www.houghton.edu\/business\/houghton-consulting-group\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/Business-Class-students.jpg","keywords":"houghton consulting group, business, marketing, financing, finances, business research"},{"title":"Houghton Choir","link":"https:\/\/www.houghton.edu\/music\/ensembles\/houghton-choir\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/College-Choir.jpg","keywords":"choir, ensembles, ensemble"},{"title":"Honors in London","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/honors\/london\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/04\/HoughtonUniversity-LondonHonors-Bus.jpg","keywords":"honors, london, study abroad, off-campus,"},{"title":"Honors at Houghton","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/honors\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/04\/Houghton-London-Honors.jpg","keywords":"honors, high academic, first-year, london, science"},{"title":"Homeschoolers","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/homeschoolers\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/DSC_0152.JPG-Website-Header.jpg","keywords":"homeschool, homeschoolers, homeschool family, homeschool families"},{"title":"History Major","link":"https:\/\/www.houghton.edu\/humanities\/history-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University_Business-Class-2019.jpg","keywords":"history, adolescence, high school, museum, teacher, history teacher"},{"title":"History","link":"https:\/\/www.houghton.edu\/about\/history\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/09\/Houghton-History-Classroom.jpg","keywords":"institution history, college history, university history"},{"title":"Highlander Wilderness Adventure","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/highlander-wilderness-adventure\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-Highlander-Wilderness-Adventure-6.jpg","keywords":"highlander wilderness adventure, wilderness, orientation, adventure"},{"title":"Highlander Passport Day","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/highlander-passport-day\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/04\/240325-AcceptedStudentsDay-039-Website-Header.webp","keywords":"accepted students, new students, move in, move-in, orientation"},{"title":"Highlander Open House","link":"https:\/\/www.houghton.edu\/visit\/highlander-open-house\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/10\/HoughtonUniversity_PassportDay.jpg","keywords":"open house, preview day"},{"title":"Highlander Club","link":"https:\/\/www.houghton.edu\/giving\/giving-societies\/highlander-club\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-MW100822HC_0415.jpg","keywords":"giving, giving clubs"},{"title":"High School Counselors","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/high-school-counselors\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Mascot-Photo-Shoot-Countdown-Website-Header-1.jpg","keywords":"high school, counselors, admission"},{"title":"Health & Wellness","link":"https:\/\/www.houghton.edu\/health\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-University-230318-FEARLESS-Ronnie-Lott-5.jpg","keywords":"pre-health, health, medical, sport, recreation and wellness major, equestrian studies, equine studies, equine therapy, pre-med, pre-nursing, pre-vet, pre-veterinary, pre-physical therapy, pre-pt, pre-pharmacy, biomedical sciences, exercise science"},{"title":"Guidelines","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/off-campus-studies\/guidelines\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Tanzania-2016.jpg","keywords":"off-campus studies, abroad, study abroad"},{"title":"Greatbatch Emerging Artists","link":"https:\/\/www.houghton.edu\/music\/greatbatch-emerging-artist\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/230110-MusicRecordingStudio1-Website-Header.jpg","keywords":"music, EP, recording"},{"title":"Graphic Design","link":"https:\/\/www.houghton.edu\/art\/studio-art-major\/graphic-design\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/04\/Houghton-Unviersity-240418-MacLab-08.webp","keywords":"art, fine arts, studio, studio art, artist, fine artist, art history, graphic design, 2D design, design, package design, information graphics"},{"title":"Graduate Programs\u2014Music","link":"https:\/\/www.houghton.edu\/graduate\/music\/programs\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/music-percussion-1.jpg","keywords":"graduate, grad, music, masters, M.A., M.M."},{"title":"Graduate Music Auditions","link":"https:\/\/www.houghton.edu\/graduate\/music\/admissions\/auditions-music\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/music-Strings.jpg","keywords":"music, graduate, audition, greatbatch"},{"title":"Graduate Assistantships","link":"https:\/\/www.houghton.edu\/graduate\/music\/admissions\/assistantships\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/220505-ScienceHonors-02-Website-Header-1.jpg","keywords":"assistantships, music, greatbatch, grad, graduate"},{"title":"Graduate Admission\u2014Music","link":"https:\/\/www.houghton.edu\/graduate\/music\/admissions\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/220505-ScienceHonors-02-Website-Header-1.jpg","keywords":"music, grad, graduate, admission"},{"title":"Graduate","link":"https:\/\/www.houghton.edu\/graduate\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/05\/MBA_13-Website-Header.jpg","keywords":"graduate, grad, master, masters, MBA, MSED, business, literacy"},{"title":"Grad Party - Highlander Style","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/grad-party-checklist\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-Unviersity-Balloons-Move-In-Day.jpg","keywords":"accepted students, new students"},{"title":"Glossary of Terms","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/resources-faqs\/glossary\/","image":false,"keywords":"financial aid terms, FAFSA, TAP, PELL, grants, scholarships"},{"title":"Global Studies Major","link":"https:\/\/www.houghton.edu\/humanities\/global-studies-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/12\/Tanzania-2016.jpg","keywords":"intercultural studies, global studies, diversity, non-profit, nonprofit, social work, missionary, missions, social worker, overseas work, study abroad,"},{"title":"Giving Societies & Clubs","link":"https:\/\/www.houghton.edu\/giving\/giving-societies\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-MW101223HU_0021.jpg","keywords":"giving, giving clubs"},{"title":"Giving","link":"https:\/\/www.houghton.edu\/giving\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/luckey_memorial.jpg","keywords":"give, giving, donate, legacy giving, societies, scholarship fund, advancement"},{"title":"Future Planning","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/vocation-and-calling\/future-planning\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/Fall-2016-Website-Header.jpg","keywords":"job, career, vocation, internship"},{"title":"Friends of Greatbatch","link":"https:\/\/www.houghton.edu\/giving\/giving-societies\/friends-of-greatbatch\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/220315-Fearless-RebekahScharf-6-Music.jpg","keywords":"giving, giving clubs,"},{"title":"Founder\u2019s Promise Scholarship","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/types-of-aid\/founders-promise-scholarship\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/11\/Aerial-Historic-Website-Header.jpg","keywords":"scholarship, financial aid"},{"title":"Forms, Applications and Resources","link":"https:\/\/www.houghton.edu\/health\/pre-med\/forms-applications-and-resources\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/biology-students.jpg","keywords":"medical school, application"},{"title":"Forms","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/forms\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/01\/fancher-hall.jpg","keywords":"internships, internship, academic petition, course add\/drop, course withdrawal, diploma, graduation, honors, independent study, declare a major, transcript, withdraw, mayterm, student concern"},{"title":"Forensic Science Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/forensic-science-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/02\/Houghton-University-240105-Biology_Poythress-25.webp","keywords":"forensics, crime science, biology, biomedical sciences, chemistry, biochemistry, criminal justice, psychology"},{"title":"Find Your Admission Counselor","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/find-your-counselor\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/admission_team_cropped.jpg","keywords":"admission, counselor, advisor"},{"title":"Financial Validation","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/resources-faqs\/policies\/validation\/","image":false,"keywords":"financial aid, aid, applying for financial aid, award letter, student financial services"},{"title":"Financial Aid Resources and FAQs","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/resources-faqs\/","image":false,"keywords":"financial aid, resources"},{"title":"Financial Aid Policies","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/resources-faqs\/policies\/","image":false,"keywords":"financial aid, aid eligibility, academic progress, transfer credits"},{"title":"Financial Aid & Ability to Benefit\u2014Homeschoolers","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/homeschoolers\/financial-aid\/","image":false,"keywords":"homeschool, homeschoolers, financial aid"},{"title":"Fee Schedule","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/fee-schedule\/","image":false,"keywords":"fees, room, board, tuition, honors fees"},{"title":"Federal Aid","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/types-of-aid\/federal-aid\/","image":false,"keywords":"federal aid, financial aid, scholarships, grants, work study"},{"title":"Fearlessly Forward\u2014Houghton University","link":"https:\/\/www.houghton.edu\/about\/fearlessly-forward-houghton-university\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/05\/220514_Houghton-Commencement_17-Website-Header.jpg","keywords":"houghton university, university announcement, mission, vision"},{"title":"Fall Online Courses","link":"https:\/\/www.houghton.edu\/online\/online-courses\/fall-online-courses\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/fall-leaves.jpg","keywords":"online classes, early college, dual enrollment, online degree, fall classes"},{"title":"Faculty Openings","link":"https:\/\/www.houghton.edu\/employment\/faculty-openings\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/Music-Inudstry-2019-Website-Header.jpg","keywords":"employment, jobs"},{"title":"Faculty & Staff\u2014Music","link":"https:\/\/www.houghton.edu\/graduate\/music\/faculty-staff\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/220829-Convocation-08_Kevin_Dibble.jpg","keywords":"faculty, staff, professors, administration"},{"title":"Faculty & Staff","link":"https:\/\/www.houghton.edu\/faculty-staff\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/08\/Houghton-University-230822-LaunchDay_FacultyStaff.jpg","keywords":"faculty, staff, professors, administration"},{"title":"Experiential Learning","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/experiential-learning\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/04\/Houghton-Tanzania-2018.jpg","keywords":"experiential learning, research, off-campus study, study abroad, internships"},{"title":"Exercise Science Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/exercise-science-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/03\/Houghton-University-DSC05710.webp","keywords":"exercise science, activity, cardio, sports science, sports medicine, pt, pre-physical therapy, physical therapy, athletics, trainer, athletic trainer, personal trainer, fitness, fitness training, fitness trainer, coach, physiology, kinesiology, kinesiologist, occupational therapy, OT,"},{"title":"Equestrian Wish List","link":"https:\/\/www.houghton.edu\/health\/equestrian-studies\/wish-list\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-Equestrian-web-IMG_5526.webp","keywords":"equestrian studies"},{"title":"Equestrian Summer Camp","link":"https:\/\/www.houghton.edu\/summer-camps\/equestrian-camp\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/11\/Houghton_Equestrian.jpg","keywords":"equestrian, camp, summer"},{"title":"Equestrian Studies FAQ","link":"https:\/\/www.houghton.edu\/health\/equestrian-studies\/faqs\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-Univesity-Equestrian-web-IMG_5189.webp","keywords":"equestrian studies"},{"title":"Equestrian Studies","link":"https:\/\/www.houghton.edu\/health\/equestrian-studies\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-230405-FEARLESS-Cassidy-Kuhlmann-13.webp","keywords":"equestrian, equestrian management, equestrian performance, horses, equine, equestrian, riding, showmanship, dressage, english riding, western riding, training, ponies, tack, hippotherapy, equine-assisted therapy, therapeutic riding, riding therapy, horse show"},{"title":"Equestrian Lesson Horses","link":"https:\/\/www.houghton.edu\/health\/equestrian-studies\/lesson-horses\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-Equestrian-IMG_3262-Web.webp","keywords":"equestrian horses, equestrian"},{"title":"Equestrian Facilities","link":"https:\/\/www.houghton.edu\/health\/equestrian-studies\/facilities\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-MW101223HU_0047.jpg","keywords":"equestrian center"},{"title":"Equestrian Events","link":"https:\/\/www.houghton.edu\/health\/equestrian-studies\/events\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-MW100822HC_0025.webp","keywords":"equestrian events"},{"title":"EPIC Ropes Course & Zipline Information","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/epic-adventures\/ropes-course\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-230623-ZipLine-8-WebP.webp","keywords":"epic adventures, ropes course, zipline"},{"title":"EPIC Facilities","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/epic-adventures\/facilities\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-230623-RopesCourse-CarterSisson-5.jpg","keywords":"ropes course, zipline, EPIC adventures, hiking"},{"title":"EPIC Adventures","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/epic-adventures\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/canoe.jpg","keywords":"epic adventures, adventure, hiking, canoeing, trails, ropes course, ski hill,"},{"title":"Environmental Science Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/environmental-science-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/Biology-2016_stream.jpg","keywords":"environmental science, environment, climate change, environmentalism, stewardship, sustainability, alternative energy, environmental studies, natural resources, creation care, conservation"},{"title":"Ensembles","link":"https:\/\/www.houghton.edu\/music\/ensembles\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Wind-Ensemble-flute.jpg","keywords":"ensembles, chamber"},{"title":"English Major","link":"https:\/\/www.houghton.edu\/humanities\/english-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/English-Class.jpg","keywords":"english, writing, language, composition, literature, classics, poetry, prose, pr, journalism, teacher, copywriter, content writer, journalist, social media coordinator, SEO specialist, proposal writer, editorial, technical writer, copy editor"},{"title":"ENCORE","link":"https:\/\/www.houghton.edu\/encore\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/220824-Move-In-chapel.jpg","keywords":"senior citizens, seniors"},{"title":"Employment","link":"https:\/\/www.houghton.edu\/employment\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/08\/Houghton-University-230822-LaunchDay_FacultyStaff.jpg","keywords":"employment, jobs, hiring, employees, faculty position, staff positions, administrative jobs, careers"},{"title":"Employers","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/vocation-and-calling\/employers\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/03\/Houghton-University-FEARLESS-Malachi-Yeager-04.webp","keywords":"employer, hiring, internships, job"},{"title":"Email and Student ID Setup","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/technology-email-student-id\/email-and-student-id-setup\/","image":false,"keywords":"student email, student ID, tech, technology"},{"title":"Education","link":"https:\/\/www.houghton.edu\/education\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-University-220901-Fearless-Nayellis-2.jpg","keywords":"education, inclusive education, music education, adolescence education, childhood education, inclusive"},{"title":"Early College\/Dual Enrollment","link":"https:\/\/www.houghton.edu\/early-college\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/02\/Students-Computers-Online.jpg","keywords":"early college, high school,"},{"title":"Drawing","link":"https:\/\/www.houghton.edu\/art\/studio-art-major\/drawing\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/02\/Watercolor-Class-2016-Website-Header.jpg","keywords":"art, fine arts, studio, studio art, artist, fine artist, art history, drawing, painting, watercolor, pastel, figure drawing, charcoal, abstract drawing, art teacher, art education"},{"title":"Donate a Horse","link":"https:\/\/www.houghton.edu\/health\/equestrian-studies\/wish-list\/donate-a-horse\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/03\/Houghton-University-Equestrian-horse-field.webp","keywords":"equestrian, equine"},{"title":"Dining Options","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/residence-life\/dining\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/Java-101-2019.jpg","keywords":"dining, metz"},{"title":"Digital Media Marketing Concentration","link":"https:\/\/www.houghton.edu\/business\/communication-major\/digital-media-marketing\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Business-Houghton.jpg","keywords":"integrated marketing, communication, concentration, communication, communications"},{"title":"Deciding Students","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/deciding-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Business-Class-student-listening.jpg","keywords":"undecided, deciding, undeclared, exploratory"},{"title":"Data Science Major","link":"https:\/\/www.houghton.edu\/math-science\/data-science-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Data-Science_computer.jpg","keywords":"data, data science, ai, machine learning, business analytics, business intelligence, statistics, data analytics, algorithms, big data, data management, data mining, data security, data visualization, deep learning, cloud computing, predictive analytics, sports analytics, data privacy, data for social good, cybersecurity, data science analytics, programming, artificial intelligence,"},{"title":"Cybersecurity Concentration","link":"https:\/\/www.houghton.edu\/math-science\/cybersecurity\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/09\/Houghton-University-student-laptop.jpg","keywords":"network and system security, cyber forensics, ethical hacking"},{"title":"Current Students","link":"https:\/\/www.houghton.edu\/current-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/06\/Houghton-University-230315-Hat-Photos-10.jpg","keywords":"current students, students, moodle, self service, webmail, student handbook, student account center, gmail"},{"title":"Current Projects","link":"https:\/\/www.houghton.edu\/giving\/current-projects\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-Aerial-Equestrian-Center-MW101223HU_0047.jpg","keywords":"giving, endowed funds, endowed chair, endowed programs, equestrian center, athletic capital projects"},{"title":"Criminal Justice Major","link":"https:\/\/www.houghton.edu\/social-sciences\/criminal-justice-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/student-on-laptop.jpg","keywords":"criminal justice, investigation, law, enforcement, police, fbi, federal, law enforcement, justice, cop, corrections, pre-law, lawyer, attorney, public defender, officer"},{"title":"CPT Employment","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/intercultural-programs\/employment-non-citizen\/cpt\/","image":false,"keywords":"international students, curricular practical training, CPT"},{"title":"COVID-19","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/health-services\/covid-19\/","image":false,"keywords":"covid, coronavirus, covid-19, covid 19, masking, vaccine,"},{"title":"Courses\u2014MSED","link":"https:\/\/www.houghton.edu\/graduate\/literacy-education\/msed-courses\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/01\/MSED-Teacher.jpg","keywords":"MSED, grad, graduate, online, literacy"},{"title":"Courses\u2014MBA","link":"https:\/\/www.houghton.edu\/graduate\/master-of-business-administration\/mba-courses\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/08\/MBA-Conference-Room.jpg","keywords":"master of business administration, mba, business, finance, leadership, management, ceo, cfo, cmo, executive,"},{"title":"Course Selection","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/course-selection\/","image":false,"keywords":"course preference, course selection, courses, new students"},{"title":"Course Preference Form","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/new-students\/course-selection\/course-preference-form\/","image":false,"keywords":"course preference, classes, courses"},{"title":"Counseling Services","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/counseling\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/HoughtonUniversity_Chamberlain-Center.jpg","keywords":"counseling services, center for student success, counseling"},{"title":"Contact Financial Aid Staff","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/contact\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Fancher-Hall.jpg","keywords":"financial aid support, financial aid staff"},{"title":"Confirm Your Enrollment","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/confirm-enrollment\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/01\/DSC_0077.JPG-Website-Header-1.jpg","keywords":"deposit, confirm, decision, may 1"},{"title":"Conducting, M.M.","link":"https:\/\/www.houghton.edu\/graduate\/music\/programs\/conducting-m-m\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/student-conducting_music.jpg","keywords":"music, musical, worship, lesson, audition, choir, chorus, band, wind ensemble, orchestra, symphony, conducting, performances, concert, aria, concerto competition, recital, music theory, music history, global music, world music, master's in music, music master's, graduate music,"},{"title":"Computer Science Major","link":"https:\/\/www.houghton.edu\/math-science\/computer-science-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Computer-Science_student-at-board.jpg","keywords":"computer science, networking, information technology, it, programming, software, IS, database management, technology, software developer web developer, cybersecurity consultant, IT consultant, multimedia programmer, database administrator"},{"title":"Composition, M.M.","link":"https:\/\/www.houghton.edu\/graduate\/music\/programs\/composition-m-m\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/sheet-music.jpg","keywords":"music, musical, worship, lesson, audition, music theory, music history, global music, world music, composition, composer, writing music, aria, concerto, film music, master's in music, music master's, graduate music,"},{"title":"Compare the Costs of College","link":"https:\/\/www.houghton.edu\/out-of-pocket-calculator\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/220514_Houghton-Commencement.jpg","keywords":"college costs, financial aid, tuition"},{"title":"Commuter Students","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/commuter-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/03\/HoughtonUniversity_230306-Commuter-Lounge.jpg","keywords":"commuter, commuting, admission"},{"title":"Community Recreation & Events","link":"https:\/\/www.houghton.edu\/community\/","image":false,"keywords":"EPIC adventures, KPAC, music performances, Ortlip Art Gallery, exhibitions, fitness facilities, ENCORE"},{"title":"Communication Major","link":"https:\/\/www.houghton.edu\/business\/communication-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/students-classroom-houghton.jpg","keywords":"communication, photography, photographer, video, videographer, filmmaker, communicator, designer, adobe, avid, final cut, fcpx, marketer, marketing, speaking, presentation, presenter, media, production, journalism, digital media, media production, broadcasting, journalism, advertising, public relations, PR, rhetoric"},{"title":"Commencement","link":"https:\/\/www.houghton.edu\/commencement\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-Commencement-2023_353.webp","keywords":"commencement, graduates, graduating, 2022, graduation, class of 2022"},{"title":"College and University Partners","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/degree-partners\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/02\/220518-VOCA-InterviewRoom.jpg","keywords":"special admission, pre-health, chiropractic, nursing, pharmacy, dentistry, medicine"},{"title":"Collaborative Performance, M.M.","link":"https:\/\/www.houghton.edu\/graduate\/music\/programs\/collaborative-performance-m-m\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Vocal-Recital-piano-vocal.jpg","keywords":"music, musical, worship, lesson, audition, choir, chorus, band, wind ensemble, orchestra, symphony, strings, brass, woodwind, percussion, drums, voice, piano, organ, guitar, flute, clarinet, saxophone, oboe, bassoon, trumpet, horn, trombone, euphonium, baritone, tuba, drums, violin, viola, cello, bass, soprano, alto, tenor, performance, collaborative piano, jazz, A cappella, performances, concert, aria, concerto competition, recital, harpsichord, master's in music, music master's, graduate music,"},{"title":"Class Scholarship Leaderboard","link":"https:\/\/www.houghton.edu\/giving\/class-scholarship-leaderboard\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/220824-Move-In-Luckey.jpg","keywords":"giving, scholarship, learderboard"},{"title":"Class Reunions","link":"https:\/\/www.houghton.edu\/alumni\/class-reunions\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/03\/MW100822HC_0368-scaled.jpg","keywords":"class reunions, homecoming, reunion, alumni"},{"title":"Chemistry Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/chemistry-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/03\/Houghton-University-220614-Shannon-Summer-Research-hood-1.webp","keywords":"chemistry, chemist, science, health, medicine, research, organic chemistry, physical chemistry, analytical chemistry, forensics"},{"title":"Chamber Ensembles","link":"https:\/\/www.houghton.edu\/music\/ensembles\/chamber\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Wind-Ensemble-percussion.jpg","keywords":"chamber music, ensemble, crass, percussion, woodwinds, strings"},{"title":"Ceramics","link":"https:\/\/www.houghton.edu\/art\/studio-art-major\/ceramics\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Ceramics-Class-Header.jpg","keywords":"art, bfa, fine arts, studio, studio art, artist, fine artist, ceramics, pottery, 3d art, book arts, art teacher, art education, potter"},{"title":"Center for Student Success","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/campuslife.jpg","keywords":"academic support, counseling, counseling services, accessibility services, writing center, writing support, voca, office of vocation and calling, career center"},{"title":"Center for Data Science Analytics","link":"https:\/\/www.houghton.edu\/about\/centers-and-institutes\/center-for-data-science-analytics\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Data-Science_super-bowl.jpg","keywords":"data science, analytics, sport analytics"},{"title":"CCCU Opportunities","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/off-campus-studies\/cccu-programs\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/03\/Houghton_Tanzania.jpg","keywords":"cccu programs, off-campus studies, study abroad"},{"title":"Catalog","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/academics\/catalog\/","image":false,"keywords":"catalog, course catalogue, academic catalog"},{"title":"Career Exploration","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/vocation-and-calling\/career-exploration\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/02\/Houghton-University-230508-Students-outside-14.webp","keywords":"career, vocation, calling, coaching"},{"title":"Canadian Students","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/canadian-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/Fall-Students-2016-Website-Header.jpg","keywords":"canada, canadian,"},{"title":"Canada Teacher Certification","link":"https:\/\/www.houghton.edu\/education\/canada-teacher-certification\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/03\/Houghton-University-240318-Education-17.webp","keywords":"teaching, education, Canada, Canadian, teaching certification, certification"},{"title":"Campus Map & Directions","link":"https:\/\/www.houghton.edu\/visit\/campus-map\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/About-Aerial_1.jpg","keywords":"map, campus, houghton,"},{"title":"Campus Employment","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-services\/payroll\/campus-employment\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/java-101_student.jpg","keywords":"student employment, on-campus job, campus jobs, campus employment"},{"title":"Calendars & Important Dates","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/calendars-important-dates-and-daily-schedules\/","image":false,"keywords":"calendar, dates, semester, academic calendar, start, housing calendar, important dates, daily schedule, class schedule"},{"title":"Business Administration Major","link":"https:\/\/www.houghton.edu\/business\/business-administration-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/12\/Business-Class-2019-Website-Header.jpg","keywords":"business administration, business, management, sales, management, marketing, finance, economics, entrepreneur, operations, accounting, finance, international business, international economics, statistics, non-profit, NGO, consultant, consulting"},{"title":"Business & Communication","link":"https:\/\/www.houghton.edu\/business\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/07\/Houghton-University-230216-Fearless-Johanna-Borges-2.jpg","keywords":"business, communication, business administration, accounting, human resources management, organizational management, marketing"},{"title":"Buffalo, NY","link":"https:\/\/www.houghton.edu\/undergraduate\/buffalo\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Buffalo-East-Students-15-Website-Header.jpg","keywords":"buffalo, king urban life center, extension sites, western ny, western new york, associate, 2 year, two year"},{"title":"Bonus Year Application","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/houghton-promise\/bonus-year-application\/","image":false,"keywords":"bonus year"},{"title":"Board of Trustees","link":"https:\/\/www.houghton.edu\/about\/president\/board-of-trustees\/","image":false,"keywords":"bot, trustees"},{"title":"Biomedical Sciences Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/biomedical-sciences-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/03\/Houghton-University-240105-Biology_Hammers-24.webp","keywords":"biomedical sciences, pre-health, pre-med, pre-"},{"title":"Biology Research","link":"https:\/\/www.houghton.edu\/natural-sciences\/biology-research\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/07\/Greenhouse.jpg","keywords":"biology research"},{"title":"Biology Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/biology-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/02\/Houghton-University-240105-Biology_Poythress-33-3.webp","keywords":"biology, biologist, science, research science, health, medicine, biology, outdoors, science, natural science, pre-med, research, ornithology, forestry, environmental science, bio, genetics, microbiology, ecology"},{"title":"Biology Facilities","link":"https:\/\/www.houghton.edu\/natural-sciences\/biology-facilities\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-230428-Biopac-2.jpg","keywords":"biology facilities, biology equipment, Paine Center for Science, labs, laboratories, greenhouse, biopac"},{"title":"Biochemistry Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/biochemistry-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/03\/Houghton-Unviersity-220614-Shannon-Summer-Research.webp","keywords":"biochemistry, biochemist, research science, medical school, medicine, health, pharmacist, science, biochemistry, biochem, science, natural science, chemistry, biology, pharmacy, pre-pharmacy, chemist, biomedical, biotechnologist, molecular, immunology"},{"title":"Bible & Theology Major","link":"https:\/\/www.houghton.edu\/ministry\/bible-theology-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/12\/Houghton-University-240321-FEARLESS-ElijahSproull_3.webp","keywords":"bible, theology, biblical studies, theological studies, ministry, pastoral care, missions, counseling, youth ministries, pastoral ministry, sports ministry, chaplaincy, youth minister, youth pastor, bible teacher"},{"title":"Behavioral Neuroscience Major","link":"https:\/\/www.houghton.edu\/social-sciences\/behavioral-neuroscience-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/psychology.jpg","keywords":"Neuroscience, biopsychology, behavioral neuroscience, psychobiology, brain studies, cognitive neuroscience, brain, behavior"},{"title":"Basketball Summer Camp","link":"https:\/\/www.houghton.edu\/summer-camps\/basketball-camp\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/02\/Mens-Basketball-2022-23-Action-Shots-Website-Header.jpg","keywords":"basketball, camp, summer"},{"title":"Audit Sheets 2023-2024","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/academic-information\/audit-sheets-2023-2024\/","image":false,"keywords":"registrar, major requirements"},{"title":"Audit Sheets 2022-2023","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/academic-information\/audit-sheets-2022-2023\/","image":false,"keywords":"registrar, major requirements"},{"title":"Audit Sheets 2021-2022","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/academic-information\/audit-sheets-2021-2022\/","image":false,"keywords":"registrar, major requirements"},{"title":"Audit Sheets 2020-2021","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/academic-information\/audit-sheets-2020-2021\/","image":false,"keywords":"registrar, major requirements"},{"title":"Audit Sheets 2019-2020","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/academic-information\/audit-sheets-2019-2020\/","image":false,"keywords":"registrar, major requirements"},{"title":"Audit Sheets 2018-2019","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/academic-information\/audit-sheets-2018-2019\/","image":false,"keywords":"registrar, major requirements"},{"title":"Associate of Science in Psychology","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/programs\/associate-of-science-in-psychology\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/Student-Life-2016-Website-Header-1.jpg","keywords":"associates degree, aa, psychology, online"},{"title":"Associate of Science in Business","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/programs\/business\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/Business-Investment-Center-1.jpg","keywords":"associate, business, a.s., online"},{"title":"Associate of Arts","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/programs\/associate-of-arts\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/02\/Online-Top-Tips.jpg","keywords":"associates degree, aa, liberal arts, online"},{"title":"Arts & Culture","link":"https:\/\/www.houghton.edu\/community\/arts-culture\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/09\/Houghton-University-Senior-Art-Show.jpg","keywords":"artist series, ortlip gallery, art exhibits, arts, culture, music performances, concerts, performances"},{"title":"Art Facilities & Equipment","link":"https:\/\/www.houghton.edu\/art\/facilities-equipment\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/11\/Stevens-Art-Studios.jpg","keywords":"art studios, facilities, equipment, digital, printmaking, lighting, mac lab, ceramics, sculpture, furniture, design, darkroom, painting, drawing, photography"},{"title":"Art","link":"https:\/\/www.houghton.edu\/art\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/04\/HoughtonUniversity_Pastels-02.jpg","keywords":"art, studio art, applied design and visual communication, graphic design"},{"title":"Applying to Honors","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/honors\/apply\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/08\/Big-Ben-13-Honors.jpg","keywords":"honors program, london honors, science honors"},{"title":"Applying for Aid","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/applying-for-aid\/","image":false,"keywords":"apply for financial aid, financial aid"},{"title":"Applying","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/apply\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/05\/Spring-2016-Website-Header_flipped.jpg","keywords":"apply, applying, admission, houghton, ny"},{"title":"Apply","link":"https:\/\/www.houghton.edu\/apply\/","image":false,"keywords":"apply, application, submit,"},{"title":"Applied Physics Major","link":"https:\/\/www.houghton.edu\/natural-sciences\/applied-physics-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/11\/221027-HoughtonUniversity_PhysicsLab.jpg","keywords":"engineering, electrical, modeling, architecture, electronic, circuit, logistics, technical, ECE, robotics, cyber security, networking, signal processing, energy systems, microelectronics, nanotechnology, systems design, control systems, power systems, smart power grids,"},{"title":"Applied Design & Visual Communication Major","link":"https:\/\/www.houghton.edu\/art\/applied-design-visual-communication-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/04\/Houghton-Unviersity-240419-MacLab-03.webp","keywords":"bfa, fine arts, adobe, printing, video, photography, camera, photographer, designer, artist, videographer, filmmaker, creative, illustrator, indesign, production, typography, art, design, graphic design, digital arts, photography, animation, film, film production, sound design, editing, photoshop, adobe, social media, influencer, media, editor, freelance, illustrator, photojournalism, blogging, vlogging, youtuber, director, ads, advertising, marketing, app design, web design, art history, applied design, visual communication"},{"title":"Applications and Forms","link":"https:\/\/www.houghton.edu\/undergraduate\/tuition-aid\/applying-for-aid\/apps-forms\/","image":false,"keywords":"financial aid, FAFSA"},{"title":"Alumni Success and Career Outcomes","link":"https:\/\/www.houghton.edu\/alumni-success-and-career-outcomes\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Commencement-2017-Website-Header.jpg","keywords":"alumni, outcomes, careers, class of 2021, grad, graduates"},{"title":"Alumni Class Challenge","link":"https:\/\/www.houghton.edu\/houghtongives\/alumni-class-challenge\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/01\/220824-Move-In-Luckey.jpg","keywords":"alumni class, challenge, giving, give"},{"title":"Alumni Board","link":"https:\/\/www.houghton.edu\/alumni\/alumni-board\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/Campus-Bridge.jpg","keywords":"alumni, alumni board"},{"title":"Alumni Ambassadors","link":"https:\/\/www.houghton.edu\/alumni\/alumni-ambassadors\/","image":false,"keywords":"alumni"},{"title":"Alumni","link":"https:\/\/www.houghton.edu\/alumni\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/03\/alumni-header.jpg","keywords":"alumni, alumnus, alumnae, alum, ambassador, reunion"},{"title":"Adolescence Education Major","link":"https:\/\/www.houghton.edu\/education\/adolescence-education-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/03\/Student-Teaching-Houghton-College-1.jpg","keywords":"education, teacher, teaching, high school, adolescence, certification, certificate,"},{"title":"Admission\u2014Online","link":"https:\/\/www.houghton.edu\/undergraduate\/online\/admission\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/09\/student_online.jpg","keywords":"admission, online, undergraduate"},{"title":"Admission\u2014MSED","link":"https:\/\/www.houghton.edu\/graduate\/literacy-education\/admissions\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/01\/MSED-Teacher.jpg","keywords":"admission"},{"title":"Admission\u2014MBA","link":"https:\/\/www.houghton.edu\/graduate\/master-of-business-administration\/admissions\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/08\/MBA-Conference-Room.jpg","keywords":"admission"},{"title":"Admission\u2014Houghton, NY","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/11\/220923-Fearless-MarceloGarcia.jpg","keywords":"admission. houghton, ny"},{"title":"Administrative & Staff Openings","link":"https:\/\/www.houghton.edu\/employment\/administrative-staff-openings\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2023\/05\/Houghton-University-230325-Accepted-Students-Day-37.jpg","keywords":"jobs, careers, employment, staff, administrative, administration, openings"},{"title":"Accreditation","link":"https:\/\/www.houghton.edu\/about\/accreditation\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/Middle-States-Kickoff-Luncheon-Website-Header.jpg","keywords":"accreditation, middle states, msche, nasm, aaqep"},{"title":"Accounting Major","link":"https:\/\/www.houghton.edu\/business\/accounting-major\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2021\/12\/Investment-Center-2019-Website-Header.jpg","keywords":"accounts, budget, audit, bank, tax accountant, finance, accounting, banking, CPA, accountant, investing, auditing, non-profit, CMA, tax law, analyst, bookkeeper, financial analyst, financial consulting, consultant, business analyst, auditor, CFO,"},{"title":"Accepted Students","link":"https:\/\/www.houghton.edu\/undergraduate\/admission\/accepted-students\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/accepted-students.jpg","keywords":"accepted, new students, deposit, confirm enrollment, enroll"},{"title":"Academics","link":"https:\/\/www.houghton.edu\/undergraduate\/majors\/academics\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/Classroom-2016-Website-Header-1.jpg","keywords":"academics, departments, schools, general education"},{"title":"Academic Support and Accessibility Services","link":"https:\/\/www.houghton.edu\/undergraduate\/student-life\/student-success\/academic-support-and-accessibility\/","image":false,"keywords":"accommodations, accommodation, study advisement, tutoring, disability services, first gen, first generation"},{"title":"Academic Information","link":"https:\/\/www.houghton.edu\/current-students\/registrar\/houghton-ny-undergraduate-students\/academic-information\/","image":false,"keywords":"audit sheet, undergraduate, academic information"},{"title":"About","link":"https:\/\/www.houghton.edu\/about\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2022\/06\/First-Week-of-Classes-Fall-2019-Website-Header-2.jpg","keywords":"about, history, facts, by the numbers"},{"title":"1883 Society","link":"https:\/\/www.houghton.edu\/giving\/giving-societies\/1883-society\/","image":"https:\/\/www.houghton.edu\/wp-content\/uploads\/2024\/01\/Houghton-University-MW101223HU_0034.jpg","keywords":"giving society, giving"}]